Reducing Waste: How Upholstery Cleaning Extends Furniture Life
 Every year, millions of furniture pieces end up in landfills. Sofas, chairs, and ottomans are discarded, often prematurely, contributing to environmental strain. But what if we could keep furniture in use longer? Upholstery cleaning offers a practical solution. By maintaining and refreshing furniture, it extends usability, reduces waste, and aligns with eco-friendly living. This approach not only saves money but also supports a healthier planet.
Every year, millions of furniture pieces end up in landfills. Sofas, chairs, and ottomans are discarded, often prematurely, contributing to environmental strain. But what if we could keep furniture in use longer? Upholstery cleaning offers a practical solution. By maintaining and refreshing furniture, it extends usability, reduces waste, and aligns with eco-friendly living. This approach not only saves money but also supports a healthier planet.
Furniture takes a beating over time. Spills, dust, and pet hair build up, making sofas look worn out. Regular maintenance, like professional sofa cleaning Brisbane, can restore fabrics and prevent deterioration. Cleaning removes dirt that breaks down fibers, preserving the sofa’s structure. Consequently, a well-maintained piece can last years longer, reducing the need for replacements and keeping bulky items out of landfills.
Why Furniture Ends Up in Landfills
Many people replace furniture when it looks dirty or outdated. Stains and odors often lead to quick decisions to toss out a sofa or chair. Yet, these issues are fixable. Professional cleaning tackles deep-set stains, allergens, and smells, giving furniture a second life. Instead of adding to the 12 million tons of furniture discarded annually in the U.S. alone, cleaning helps keep pieces functional and attractive.
Moreover, replacing furniture has a hidden cost. Manufacturing new items consumes resources like wood, metal, and textiles, plus energy for production and shipping. By extending furniture life through cleaning, you cut down on this cycle of consumption. It’s a small choice with a big impact, shrinking your environmental footprint.
How Upholstery Cleaning Works
Upholstery cleaning isn’t just a surface wipe-down. Professionals use specialized tools and eco-friendly solutions to deep-clean fabrics. Steam cleaning, for instance, lifts dirt from fibers without harsh chemicals. Dry cleaning methods work well for delicate materials. These techniques revive colors, remove odors, and protect against future wear. Regular cleaning, every 12 to 18 months, keeps furniture in top shape.
But why does this matter for the environment? Landfills are overflowing, and furniture takes decades to decompose. Upholstered items, often treated with chemicals, can leach toxins into soil and water. By maintaining your furniture, you delay its trip to the dump, easing pressure on waste systems and reducing pollution.
Additional Benefits of Furniture Maintenance
Beyond environmental perks, upholstery cleaning improves your home’s health. Dust mites, pet dander, and allergens hide in fabrics, triggering allergies or asthma. A thorough cleaning removes these irritants, creating a cleaner living space. Plus, refreshed furniture looks great, boosting your home’s appeal without the cost of new pieces.
Financially, it’s a win too. Replacing a sofa can cost hundreds or thousands of dollars. Professional cleaning, by contrast, is a fraction of that price. Over time, the savings add up, letting you invest in other priorities while keeping your home stylish and sustainable.
Embracing a Sustainable Mindset
Caring for furniture reflects a broader shift toward sustainability. Small actions—like choosing to clean rather than replace—build a culture of reuse. Pair this with other habits, like buying secondhand or donating usable furniture, and the impact grows. Communities benefit when fewer items clog landfills, and future generations inherit a cleaner planet.
So, how can you start? Inspect your furniture regularly. Address spills quickly to prevent stains. Schedule professional cleanings to maintain fabric integrity. These steps keep your furniture functional and beautiful, proving that eco-friendly choices can be simple and rewarding.
READ ALSO: Sustainable Kitchen Renovations: A Step Towards Eco-Friendly Living
A Call to Action
Your furniture holds untapped potential. With proper care, it can serve your home for years while supporting environmental goals. Upholstery cleaning is more than a chore—it’s a commitment to reducing waste and living thoughtfully. Next time your sofa looks tired, don’t replace it. Clean it. The planet, and your wallet, will thank you.




 Currently, there are around 15.5 million commercial trucks plying the US roads alone, two million of which include heavy duty towing trucks and tractor trailers. All of them have massive environmental impact in terms of CO2 greenhouse gas (GHG) emissions.
Currently, there are around 15.5 million commercial trucks plying the US roads alone, two million of which include heavy duty towing trucks and tractor trailers. All of them have massive environmental impact in terms of CO2 greenhouse gas (GHG) emissions. Improvements to cut down fuel usage presented direct cost savings, whilst reducing GHG and CO2 emissions. Players in the trucking industry welcomed the recommendation since reduced costs simply means less overhead expenses that reduce their gross profits.
Improvements to cut down fuel usage presented direct cost savings, whilst reducing GHG and CO2 emissions. Players in the trucking industry welcomed the recommendation since reduced costs simply means less overhead expenses that reduce their gross profits.


 When it comes to roadside assistance, hiring the service of a dependable towing company, such as Towing Service San Jose, is imperative for you to get fast and quality service. Furthermore, you are certain that they will handle your car with utmost care throughout the process of transporting your car to the nearest mechanic in the area.
When it comes to roadside assistance, hiring the service of a dependable towing company, such as Towing Service San Jose, is imperative for you to get fast and quality service. Furthermore, you are certain that they will handle your car with utmost care throughout the process of transporting your car to the nearest mechanic in the area. Making Your Car More Eco-Friendly
Making Your Car More Eco-Friendly
 Energy use in American homes continues to rise each year, and the push for greener systems is getting stronger. Homeowners want comfort without high utility bills, and companies across Maryland are responding with smarter heating, cooling, and plumbing solutions. Frederick and nearby communities are also seeing more interest in climate friendly home upgrades. This is where trusted local services come in, including reliable heating repairs, system upgrades, and long term maintenance.
Energy use in American homes continues to rise each year, and the push for greener systems is getting stronger. Homeowners want comfort without high utility bills, and companies across Maryland are responding with smarter heating, cooling, and plumbing solutions. Frederick and nearby communities are also seeing more interest in climate friendly home upgrades. This is where trusted local services come in, including reliable heating repairs, system upgrades, and long term maintenance. Every year, millions of tons of textile waste end up in landfills, much of it coming from synthetic activewear that takes hundreds of years to decompose. Yoga wear, known for its stretch and comfort, is no exception. While yoga promotes mindfulness and balance, the clothes many practitioners wear often tell a different story, one tied to pollution, microplastics, and unsustainable production. The good news is that a new movement is emerging: eco-friendly yoga fashion designed to align our values with our wardrobe.
Every year, millions of tons of textile waste end up in landfills, much of it coming from synthetic activewear that takes hundreds of years to decompose. Yoga wear, known for its stretch and comfort, is no exception. While yoga promotes mindfulness and balance, the clothes many practitioners wear often tell a different story, one tied to pollution, microplastics, and unsustainable production. The good news is that a new movement is emerging: eco-friendly yoga fashion designed to align our values with our wardrobe.
 Every day, millions search for answers to pressing environmental questions. Climate change, sustainable practices, and eco-innovation dominate discussions, but finding reliable information can feel like searching for a needle in a haystack. The internet is flooded with data, yet much of it is outdated, biased, or simply inaccurate. For environmentalists, researchers, and students, this creates a frustrating barrier. How do you separate fact from fiction? Curated directories offer a solution, streamlining the quest for trustworthy green knowledge.
Every day, millions search for answers to pressing environmental questions. Climate change, sustainable practices, and eco-innovation dominate discussions, but finding reliable information can feel like searching for a needle in a haystack. The internet is flooded with data, yet much of it is outdated, biased, or simply inaccurate. For environmentalists, researchers, and students, this creates a frustrating barrier. How do you separate fact from fiction? Curated directories offer a solution, streamlining the quest for trustworthy green knowledge. As energy efficiency becomes more important, the construction industry continues to look for better building materials. One of the standout innovations in recent years is vacuum insulating glass (VIG), the most efficient glass. This advanced type of glass is designed to reduce heat transfer, helping buildings stay cooler in summer and warmer in winter. It looks like regular glass, but it works much harder behind the scenes. By saving energy, it also helps reduce carbon emissions and supports a more sustainable way of living.
As energy efficiency becomes more important, the construction industry continues to look for better building materials. One of the standout innovations in recent years is vacuum insulating glass (VIG), the most efficient glass. This advanced type of glass is designed to reduce heat transfer, helping buildings stay cooler in summer and warmer in winter. It looks like regular glass, but it works much harder behind the scenes. By saving energy, it also helps reduce carbon emissions and supports a more sustainable way of living.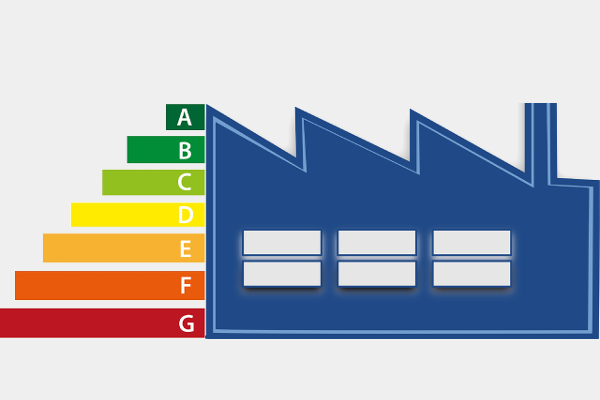



 A travel trailer cover is an essential investment for any avid traveler seeking to protect their vehicle from the elements. However, just purchasing a high-quality cover is not enough; regular maintenance is crucial in ensuring that it lasts for years to come. By taking the time to care for your cover, you can prevent wear and tear from inclement weather, dirt, and UV rays, extending its lifespan and safeguarding your investment.
A travel trailer cover is an essential investment for any avid traveler seeking to protect their vehicle from the elements. However, just purchasing a high-quality cover is not enough; regular maintenance is crucial in ensuring that it lasts for years to come. By taking the time to care for your cover, you can prevent wear and tear from inclement weather, dirt, and UV rays, extending its lifespan and safeguarding your investment. Water resistance is another critical characteristic of an effective travel trailer cover. Waterproof travel trailer covers for rain is a must! A cover that can repel water ensures that rain and snow won’t seep through, potentially causing mold, mildew, or other forms of deterioration in your trailer’s structure. It’s also beneficial to look for covers with a waterproof backing, as this additional layer reinforces the cover’s ability to seal out moisture while allowing any trapped condensation within to escape, thus preventing rust and corrosion.
Water resistance is another critical characteristic of an effective travel trailer cover. Waterproof travel trailer covers for rain is a must! A cover that can repel water ensures that rain and snow won’t seep through, potentially causing mold, mildew, or other forms of deterioration in your trailer’s structure. It’s also beneficial to look for covers with a waterproof backing, as this additional layer reinforces the cover’s ability to seal out moisture while allowing any trapped condensation within to escape, thus preventing rust and corrosion.



 However, levels of business travel stress vary from person to person as some may have had past experiences that lessen their tolerance for new and unexpected challenges; oftentimes leading to fears of unexpected events. Actually, even if seasoned business travellers are no longer apprehensive over possible negative outcomes, their body still develops tenseness that can affect their physical and mental conditions when attending critical business meetings.
However, levels of business travel stress vary from person to person as some may have had past experiences that lessen their tolerance for new and unexpected challenges; oftentimes leading to fears of unexpected events. Actually, even if seasoned business travellers are no longer apprehensive over possible negative outcomes, their body still develops tenseness that can affect their physical and mental conditions when attending critical business meetings.





 Infrastructure environment monitoring therefore is all about keeping round-the-clock visuals and tracking the ambience of remote infrastructure sites. The purpose of which is to immediately address or prevent any backend issues that could affect the availability and performance of IT functions and services.
Infrastructure environment monitoring therefore is all about keeping round-the-clock visuals and tracking the ambience of remote infrastructure sites. The purpose of which is to immediately address or prevent any backend issues that could affect the availability and performance of IT functions and services. The monitors also maintain external dry connections to water, smoke and vibration detectors or sensors. Such devices provide visibility to the ambient status of the environment.
The monitors also maintain external dry connections to water, smoke and vibration detectors or sensors. Such devices provide visibility to the ambient status of the environment. First off, the 24/7 home monitoring system is connected to a remote monitoring station manned by a team of security professionals. This team will access the home monitoring system, only with the permission of the home or business owners
First off, the 24/7 home monitoring system is connected to a remote monitoring station manned by a team of security professionals. This team will access the home monitoring system, only with the permission of the home or business owners

 The internet has a significant impact on the environment, and web design agency Wellington has a responsibility to create websites that are sustainable and eco-friendly. Here are some tips and best practices for creating environmentally friendly websites that are user-friendly and accessible.
The internet has a significant impact on the environment, and web design agency Wellington has a responsibility to create websites that are sustainable and eco-friendly. Here are some tips and best practices for creating environmentally friendly websites that are user-friendly and accessible. Today, social media is a potent force for advancing causes and making a difference. It has the potential to create real, meaningful change. Instagram has become a powerful tool for individuals and organizations to
Today, social media is a potent force for advancing causes and making a difference. It has the potential to create real, meaningful change. Instagram has become a powerful tool for individuals and organizations to 





